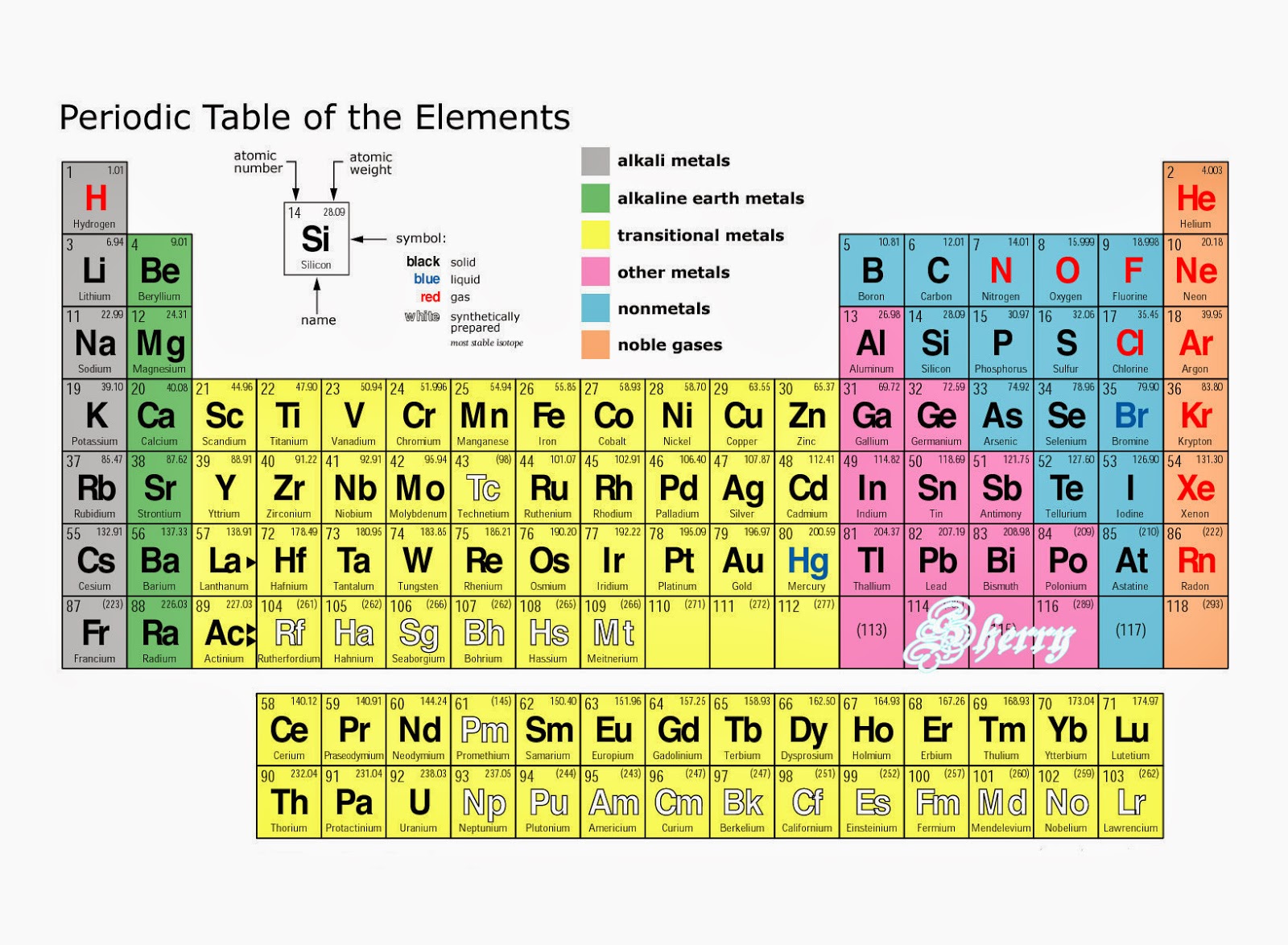
For example, the Neon or the Noble Gases group consists of elements that completely fills their octet and is in the utmost stable form. Each group of elements has different nature and attributes. Group 11: Copper or Coinage Metal GroupĪll currently known elements are divided into one of these 18 groups.Group 2: Beryllium or Alkaline Earth Metal Group.In total, there are 18 groups in a periodic table.Įach of these groups has an individual name, and the groups are named due to their nature and reactions to other elements. In general terms, a group on a periodic table is represented as the vertical columns that host elements with identical electron configuration but tends to change the nature of the elements from left to right. If you consider yourself a true connoisseur of chemistry, then it is vital that you learn about the differences between groups and periods. If you look into the periodic table, you can find that groups are arranged in a horizontal position, while on the other hand, periods are arranged in a vertical position.Īnd although they oppose each other by their arrangements and definition, both serve the purpose of arranging all known elements with proper order so that we can find them easily and efficiently.Īlso, for anyone interested in science, learning the differences between groups and periods is vital, as there is always a chance of a new element to join the ranks of the periodic table. The main difference between group and period is their position and arrangement in the periodic table. Period vs Group Periodic Table: A Brief Overview group debate, as they are the supporting pillars that make up the entire periodic table. These elements complete the seventh period or row of the periodic table.So, follow along as we take a deeper look into period vs. The Russian team that discovered element 118 named it Oganesson, symbol Og, after Yuri Oganessian, a prolific element hunter.A team of scientists from Russia and the United States named element 115, Moscovium, symbol Mc, after Moscow and element 117, Tennessine, symbol Ts, after Tennessee.Element 113 was named Nihonium, symbol Nh, proposed by Japanese researchers after the Japanese word Nihon, which means Japan.The following are the new element names and their origins: In December 2015, the IUPAC verified the existence of four new elements 113, 115, 117, and 118 and approved their addition to the periodic table. Elements up to atomic number 112 are known (not in any detail, however), and some evidence for elements 113 and above had been put forth, with elements 114 and 116 being added to the table in 2011. These synthetic elements tend to be very unstable, often existing for less than a second, so little is known about them. Larger elements (atomic numbers 101 and above) are formed by fusing nuclei of smaller elements together. Uranium has been used in this manner to produce elements 93-100. Many elements have been synthesized by bombarding known elements with sub-atomic particles such as neutrons or alpha particles. In fact, many of those seventh period elements were not known before the early twentieth century. Most recent depictions of the periodic table show an incomplete seventh period. Many periodic tables show both systems simultaneously. To eliminate confusion, the International Union of Pure and Applied Chemistry (IUPAC) decided that the official system for numbering groups would be a simple 1 through 18 from left to right. Unfortunately, there was a slightly different system in place in Europe. The first two groups are 1A and 2A, while the last six groups are 3A through 8A. The traditional system used in the United States involves the use of the letters A and B. There are two different numbering systems that are commonly used to designate groups, and you should be familiar with both. These two rows are pulled out in order to make the table itself fit more easily onto a single page.Ī group is a vertical column of the periodic table, based on the organization of the outer shell electrons. Periods 6 and 7 have 32 elements, because the two bottom rows that are separate from the rest of the table belong to those periods. 
Period 1 has only two elements (hydrogen and helium), while periods 2 and 3 have 8 elements. A new period begins when a new principal energy level begins filling with electrons. There are seven periods in the periodic table, with each one beginning at the far left. \( \newcommand\) (Credit: User:Cepheus/Wikimedia Commons Source: License: Public Domain)Ī period is a horizontal row of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
